







Content Menu
● Definition and Primary Characteristics of an Isolation Gown
>> Physical Characteristics and Coverage
● Types and Materials of Isolation Gowns
>> Classification by Protection Level
>> Material Composition and Properties
● Design and Comfort Features of Isolation Gowns
>> Ergonomics and Comfort Considerations
● Standards and Regulations Governing Isolation Gowns
>> International Standards Framework
>> Testing and Certification Processes
● Selection and Proper Use of Isolation Gowns
>> Choosing the Appropriate Isolation Gown
>> Proper Donning and Doffing Procedures
>> 1. What exactly is an isolation gown and how does it differ from a surgical gown?
>> 2. What do the different levels of isolation gowns mean?
>> 3. Can isolation gowns be reused?
>> 4. What is the proper way to put on and remove an isolation gown?
>> 5. What standards should a quality isolation gown meet?
An isolation gown is a fundamental component of personal protective equipment (PPE) designed to create a protective barrier between the wearer and potential contaminants in their environment. These specialized garments serve critical roles across numerous sectors, particularly in healthcare settings where they protect healthcare workers and patients from the transmission of infectious agents. The isolation gown has gained unprecedented visibility during global health crises such as the COVID-19 pandemic, highlighting its essential position in comprehensive infection control protocols. Understanding what constitutes an isolation gown, its various types, appropriate applications, and standards is crucial for effective implementation in any environment where protection from contamination is necessary.
The purpose of this article is to provide a comprehensive examination of isolation gowns, exploring their definition, classification, materials, design features, and appropriate usage scenarios. We will delve into the key differences between isolation gowns and other protective garments, the standards governing their manufacture, and the critical factors to consider when selecting an isolation gown for specific situations. Whether you are a healthcare professional, worker in industrial settings, or simply seeking to understand this essential protective equipment, this guide will offer valuable insights into the world of isolation gowns and their vital role in maintaining safety.
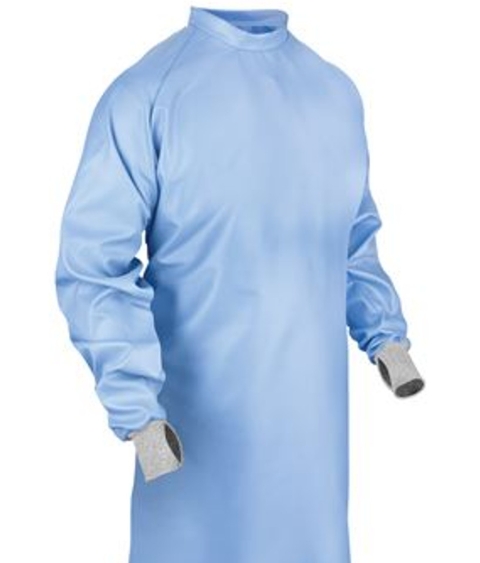
An isolation gown is specifically designed to protect the wearer's skin and clothing from the transfer of microorganisms, bodily fluids, and other potentially infectious materials in environments where such hazards exist. Unlike surgical gowns that maintain a sterile field in operating rooms, isolation gowns are primarily intended for use in isolation precautions across various settings. According to the Centers for Disease Control and Prevention's Guideline for Isolation Precautions, isolation gowns should be worn to protect healthcare workers' arms and exposed body areas during procedures and patient-care activities when anticipating contact with clothing, blood, bodily fluids, secretions, and excretions .
The isolation gown serves as a removable barrier that minimizes the transmission of pathogens between healthcare workers and patients or contaminated environments. By preventing the contamination of healthcare workers' skin and clothing, the isolation gown plays a crucial role in breaking the chain of infection transmission. This protective function extends beyond healthcare settings to include laboratories, industrial environments, and any situation where protection from hazardous substances is required.
A typical isolation gown is designed to cover the torso completely, extending from the neck to the knees or below, and the arms from the shoulder to the wrist. Most isolation gowns feature long sleeves, a open-back design with secure closures (typically ties or snaps), and varying lengths to ensure adequate protection during movement and patient care activities. The back of the isolation gown may be open or partially closed, with the coverage area depending on the specific design and intended use .
The materials used in manufacturing isolation gowns vary significantly based on the intended application and required protection level. These can range from basic non-woven fabrics like polypropylene to more sophisticated materials with enhanced fluid resistance. The design of an isolation gown must balance protection with practicality, allowing sufficient mobility for the wearer to perform necessary tasks while maintaining an effective barrier against contaminants. Modern isolation gowns often incorporate features such as elastic cuffs, reinforced critical zones, and various closure systems to enhance their protective capabilities and usability.
Isolation gowns are systematically classified according to their protective capabilities, most commonly under standards such as the Association for the Advancement of Medical Instrumentation (AAMI) PB70 classification. This system categorizes isolation gowns into four distinct levels based on their liquid barrier performance:
- Level 1 Isolation Gown: Designed for minimal risk situations with tested resistance to water only, this basic isolation gown is suitable for general patient care without anticipated fluid exposure.
- Level 2 Isolation Gown: Providing low barrier protection, this isolation gown is appropriate for situations where low fluid exposure is expected, such as during routine patient care.
- Level 3 Isolation Gown: Offering moderate barrier protection, this isolation gown is used for situations with moderate fluid exposure risks, commonly employed during procedures with higher potential for fluid contact.
- Level 4 Isolation Gown: Delivering the highest level of protection, this isolation gown is designed for situations with high fluid exposure risks, essentially impermeable and used for high-risk procedures .
Understanding these classification levels is essential for selecting the appropriate isolation gown for specific tasks and exposure risks. The selection should be based on a thorough risk assessment that considers the anticipated type and volume of fluid exposure, rather than automatically choosing the highest available protection level.
The materials used in isolation gowns significantly determine their protective properties, comfort, and environmental impact. Disposable isolation gowns are typically manufactured from various non-woven materials, with the Chinese standard GB/T 38462-2020 specifically focusing on "Textiles - Nonwoven fabrics for isolation gowns" . These materials include:
- Polypropylene: A common material for basic isolation gowns, offering reasonable protection against dry particles and limited fluid resistance.
- Spunbond-Meltblown-Spunbond (SMS): A composite material that provides enhanced fluid resistance and breathability, suitable for moderate risk situations.
- Polyethylene-coated materials: These offer higher levels of fluid protection but may compromise breathability.
- Reinforced fabrics: Some isolation gowns incorporate reinforcement in critical areas to provide additional protection where exposure risk is highest.
Reusable isolation gowns are typically constructed from woven fabrics, often cotton or cotton-polyester blends, that undergo special finishing treatments to enhance their fluid-resistant properties. These isolation gowns can be laundered and reused multiple times, potentially offering economic and environmental advantages over disposable options. However, reusable isolation gowns require proper laundering procedures to maintain their protective qualities and prevent cross-contamination .
The design of an isolation gown incorporates several important elements that enhance its protective capabilities and usability. These design features have evolved to address the practical needs of healthcare workers while maintaining effective barrier properties. Common design elements include:
- Closure systems: Isolation gowns typically feature secure closure mechanisms, most commonly tie-back systems with neck and waist ties. Some modern designs may incorporate snap fasteners or hook-and-loop closures for easier donning and doffing . The placement of these closures (typically at the back) helps minimize potential entry points for contaminants.
- Cuff designs: The cuffs of an isolation gown are critical areas where contaminants might enter. Various cuff designs exist, including elasticated cuffs, knit cuffs, and thumb loops, all aimed at ensuring a secure fit around the wrist and preventing ride-up during movement .
- Coverage variations: While standard isolation gowns cover from the neck to below the knees and the entire arms, designs may vary in their back coverage. Some offer full back coverage, while others may have partial coverage depending on the intended use and level of protection required.
- Reinforcement: Many isolation gowns incorporate reinforced sections in areas most likely to encounter contamination, such as the forearms and front torso. This reinforcement provides an additional layer of protection in high-risk zones.
These design elements are not merely functional but also contribute to the overall effectiveness of the isolation gown as protective equipment. The design must consider the range of motions and activities healthcare workers perform while ensuring continuous protection throughout use.
While protection remains the primary function of an isolation gown, comfort factors significantly influence user compliance and effectiveness. An uncomfortable isolation gown may lead to improper use, early removal, or adjustments that compromise protection. Key comfort considerations include:
- Breathability: The ability of the gown material to allow moisture vapor transmission significantly affects thermal comfort. Breathability prevents heat and moisture buildup inside the gown, which is particularly important during extended wear or in warm environments.
- Mobility: The cut and fit of an isolation gown must allow for a full range of motion without excessive stress on the materials or restrictions to movement. Design features such as raglan sleeves or strategic pleating can enhance mobility without compromising coverage.
- Weight: Lighter materials generally offer greater comfort but may sacrifice some protection. Manufacturers must balance these competing factors based on the intended use of the isolation gown.
- Closure comfort: Closure systems must be secure yet comfortable, without creating pressure points or irritation during extended wear.
The ideal isolation gown successfully balances protection with comfort, encouraging proper use while maintaining effective barrier properties. This balance becomes increasingly important during prolonged procedures or in challenging environmental conditions.

The manufacture and performance of isolation gowns are governed by various standards and regulations that ensure consistent quality and protection levels. These standards establish test methods and performance requirements that isolation gowns must meet for specific applications. Key standards include:
- AAMI PB70: The American National Standards Institute/Association for the Advancement of Medical Instrumentation liquid barrier classification standard is widely recognized and establishes the four-level classification system for isolation gowns based on their resistance to liquid penetration .
- GB/T 38462-2020: This Chinese standard specifically addresses non-woven fabrics used in isolation gowns, detailing technical requirements, test methods, and classification systems for these materials .
- European Standards: The European Union has specific standards for medical devices, including isolation gowns, under the European Medical Devices Regulation (MDR).
- FDA Regulations: In the United States, the Food and Drug Administration regulates isolation gowns as medical devices, with specific requirements depending on the intended use and classification of the gown.
These standards typically address aspects such as fluid resistance, tensile strength, breathability, and seam strength, providing a framework for evaluating the performance and safety of isolation gowns. Compliance with relevant standards helps ensure that isolation gowns provide consistent, reliable protection for users.
To verify compliance with applicable standards, isolation gowns undergo rigorous testing procedures that evaluate their protective properties. Key tests include:
- Fluid resistance testing: This evaluates the material's ability to resist liquid penetration under various conditions, often using synthetic blood or other test fluids.
- Tensile strength testing: This measures the material's resistance to tearing or breaking under stress, indicating its durability during use.
- Breathability testing: This assesses the material's resistance to moisture vapor transmission, which influences wearer comfort.
- Seam strength testing: This evaluates the durability of seams, which are potential weak points in an isolation gown.
Certification processes vary by region but generally involve assessment by recognized bodies that verify compliance with applicable standards. This may include initial testing of materials and finished products, facility inspections, and ongoing surveillance to ensure continued compliance. For manufacturers, understanding and adhering to these standards is essential for producing isolation gowns that meet the necessary safety and performance requirements.
Selecting the correct isolation gown for a specific situation requires careful consideration of multiple factors to ensure adequate protection without unnecessary discomfort or cost. The decision process should include:
- Risk assessment: Evaluate the anticipated exposure type (splash, spray, or large-volume fluid contact), potential pathogens involved, and duration of exposure. This assessment directly informs the required protection level.
- Procedure requirements: Consider the specific activities to be performed. For instance, a isolation gown used during phlebotomy with minimal fluid exposure may differ from one used during emergency trauma care with substantial fluid exposure risk.
- Comfort and mobility needs: For procedures requiring extensive movement or lasting extended periods, comfort factors such as breathability and freedom of movement become more significant.
- Environmental considerations: The choice between disposable and reusable isolation gowns may involve considering environmental impact, waste management, and life-cycle costs.
In healthcare settings, isolation gown selection is often guided by transmission-based precautions and specific procedure requirements. For standard precautions involving minimal fluid exposure, a Level 1 or 2 isolation gown may be sufficient. For contact precautions with patients known or suspected to have epidemiologically important pathogens, a higher-level isolation gown (Level 2 or 3) is typically recommended. During procedures with anticipated substantial fluid exposure, a Level 3 or 4 isolation gown provides appropriate protection .
The protective effectiveness of an isolation gown depends not only on its quality and appropriate selection but also on correct donning and doffing techniques. Improper procedures can compromise protection and lead to self-contamination. Key steps include:
Donning (Putting on):
1. Perform hand hygiene thoroughly before handling the isolation gown.
2. Unfold the gown carefully, holding it by the shoulders or neck area to avoid contact with contaminated surfaces.
3. Slip arms into the sleeves, ensuring complete coverage of the torso and arms.
4. Secure the gown at the neck and waist, ensuring all closures are properly fastened.
5. Don gloves after gowning, pulling them over the cuffs of the isolation gown to create a continuous barrier.
Doffing (Removing):
1. Remove gloves carefully using proper technique to avoid contaminating hands.
2. Perform hand hygiene after glove removal.
3. Untie or unfasten the isolation gown without touching the front exterior surfaces.
4. Pull the gown away from the body, turning it inside out during removal to contain contaminated surfaces.
5. Dispose of the used isolation gown properly according to institutional protocols.
6. Perform hand hygiene again after complete removal.
These meticulous procedures maximize the protective benefits of the isolation gown and minimize the risk of contamination during removal. Training and practice in these techniques are essential for all personnel who use isolation gowns in their work.
The isolation gown represents a critical component of personal protective equipment across numerous sectors, with particularly vital applications in healthcare settings. Understanding what an isolation gown is—its definition, classification, materials, design features, and proper usage—is essential for effective implementation in infection control and hazard protection protocols. From its fundamental role in creating a protective barrier against microorganisms and hazardous substances to its specific design considerations that balance protection with comfort, the isolation gown serves as a customizable tool that can be adapted to various risk scenarios.
The classification of isolation gowns according to standardized systems such as AAMI levels provides a framework for matching protective capabilities with specific exposure risks. Meanwhile, ongoing developments in materials science and gown design continue to enhance both the protective performance and wearer comfort of these essential garments. As global health challenges evolve and awareness of safety protocols grows, the importance of proper isolation gown selection and usage remains paramount across healthcare, laboratory, industrial, and numerous other environments.

An isolation gown is a protective garment designed to protect the wearer's skin and clothing from the transfer of microorganisms, bodily fluids, and other potentially infectious materials in environments where such hazards exist. The key differences between isolation gowns and surgical gowns lie in their intended use, sterility, and coverage areas. While isolation gowns are generally non-sterile and used for isolation precautions in various settings, surgical gowns are sterile garments specifically designed for use in surgical procedures where they must provide a critical barrier against pathogen migration in both front and back areas during sterile operations . Surgical gowns also undergo more rigorous testing for liquid barrier performance and are classified as Class II medical devices requiring regulatory approval like from the FDA.
Isolation gowns are classified into different levels based on their liquid barrier performance, most commonly under the AAMI PB70 standard which establishes four protection levels. Level 1 provides minimal barrier protection for low-risk situations with no anticipated fluid exposure. Level 2 offers low barrier protection for situations with low fluid exposure risk. Level 3 provides moderate barrier protection for situations with moderate fluid exposure risk. Level 4 delivers the highest level of protection against heavy fluid exposure and is essentially impermeable . The appropriate level should be selected based on a risk assessment considering the anticipated type and volume of fluid exposure rather than automatically choosing the highest available protection level.
The reusability of an isolation gown depends on its type and manufacturer specifications. Disposable isolation gowns are designed for single use only and should not be reused, as their protective barriers may become compromised during use. Reusable isolation gowns, typically made from woven fabrics with fluid-resistant finishes, can be laundered and reused according to manufacturer instructions . Proper laundering of reusable isolation gowns is essential to maintain their protective qualities, including specific wash cycles, detergent types, and drying parameters. During supply shortages, some facilities may implement extended use protocols for disposable isolation gowns, but these are exceptional circumstances requiring specific risk assessments and institutional policies.
Proper donning and doffing of an isolation gown is crucial to maximize its protective benefits and minimize contamination risks. For donning: (1) perform hand hygiene first; (2) unfold the gown carefully; (3) slip arms into the sleeves; (4) secure the gown at neck and waist; (5) don gloves last, pulling them over the cuffs. For doffing: (1) remove gloves carefully first; (2) perform hand hygiene; (3) untie or unfasten the gown without touching the front; (4) pull the gown away from the body, turning it inside out; (5) dispose of properly; (6) perform hand hygiene again. The key is to touch only the clean inner surfaces during removal and avoid contact with potentially contaminated outer surfaces.
A quality isolation gown should comply with relevant standards for its intended use and market. Key standards include the AAMI PB70 standard for liquid barrier classification, which establishes performance levels for isolation gowns . In China, the GB/T 38462-2020 standard specifically addresses non-woven fabrics used in isolation gowns . In the United States, the FDA regulates isolation gowns as medical devices with specific requirements. Additionally, quality isolation gowns should have clear labeling indicating their protection level, size, and intended use. When selecting isolation gowns, it's important to verify compliance with applicable standards for the specific healthcare setting or application.
[1] Centers for Disease Control and Prevention. (2020). Sequence for Putting on Personal Protective Equipment. Retrieved from CDC website.
[2] World Health Organization. (2020). Rational use of personal protective equipment for coronavirus disease (COVID-19) and considerations during severe shortages. WHO Reference Number: WHO/2019-nCov/IPC_PPE_use/2020.3
[3] GB/T 38462-2020. Textiles - Nonwoven fabrics for isolation gowns.
[4] National Medical Products Administration. Medical Device Classification.