







Content Menu
● Understanding the Rigid Bronchoscope
● Pre-Cleaning and Point-of-Use Processing
● Manual Cleaning: Step-by-Step Guide
● Disinfection and Sterilization Methods
● Drying and Storage Protocols
● Quality Control and Maintenance
>> 1. Why is manual cleaning necessary if an automated reprocessor is used?
>> 2. How often should a rigid bronchoscope undergo leak testing?
>> 3. Can the same disinfectant be used for all types of bronchoscopes?
>> 4. What is the recommended storage method for a rigid bronchoscope?
>> 5. How does the design of a rigid bronchoscope impact its cleanability?
In the field of medical visualization, proper cleaning and disinfection of bronchoscopes are critical for patient safety and instrument longevity. As a company specializing in a range of medical visualization devices, including endoscopy systems, disposable ureteroscopes, visual flexible laryngoscopes, bronchoscopy workstations, medical image processors, and video laryngoscopes, we understand the intricate processes required to maintain these sophisticated instruments. The rigid bronchoscope, specifically, demands meticulous cleaning protocols to prevent cross-contamination and ensure optimal performance. This article provides a comprehensive, step-by-step guide on how to effectively clean and disinfect a bronchoscope, incorporating industry best practices and technological advancements. We will explore both manual and automated cleaning methods, address common challenges, and highlight how our OEM services support the development of bronchoscopes that are not only technologically advanced but also designed for ease of maintenance and infection control.
The importance of properly cleaning a rigid bronchoscope cannot be overstated. Given that bronchoscopes are used in sensitive respiratory procedures, any residual biological material or pathogens can lead to serious healthcare-associated infections (HAIs). The complex structure of a rigid bronchoscope, with its long, narrow channels and intricate tips, presents unique cleaning challenges that require specialized protocols and equipment. Throughout this guide, we will emphasize the critical steps—from pre-cleaning at the point of use to final storage—that are essential for thorough decontamination. Our expertise in medical visualization informs our understanding that a clean bronchoscope is not just about patient safety; it also preserves the instrument's imaging capabilities and functional integrity, thereby supporting accurate diagnoses and effective treatments.
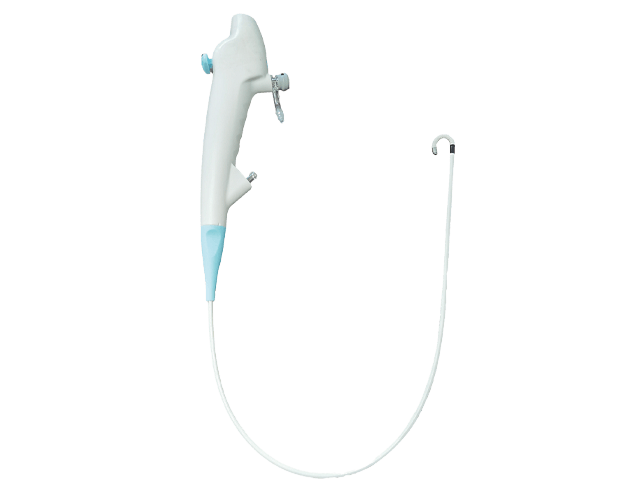
Before delving into the cleaning process, it is essential to understand the basic design and components of a rigid bronchoscope. A rigid bronchoscope is a straight, hollow metal tube used primarily for diagnostic and therapeutic procedures in the upper airways and proximal bronchi. Unlike its flexible counterpart, the rigid bronchoscope maintains its shape throughout procedures, providing stability and a larger working channel for various instruments. The key components include the main tube with a beveled distal tip, an integrated lighting system, ventilation ports, and optical telescopes. The robust construction of the rigid bronchoscope—typically made of medical-grade stainless steel or specialized alloys—ensures durability but also necessitates specific cleaning approaches to avoid damage while effectively removing contaminants.
The design of the rigid bronchoscope has evolved significantly since its inception, with modern iterations incorporating advanced optics, such as rod-lens telescopic systems and adapters for high-definition cameras. These enhancements improve visualization during procedures but add complexity to the cleaning process. For instance, the optical components are sensitive to harsh chemicals or improper handling, which can compromise image quality. Additionally, the lumen and external surfaces of the rigid bronchoscope can harbor blood, mucus, and other biological debris after use, making thorough cleaning imperative. As an OEM service provider, we design bronchoscopes with cleaning in mind, ensuring that components can be easily disassembled, accessed, and cleaned without compromising their structural or optical integrity. This proactive approach facilitates compliance with cleaning protocols and enhances the instrument's lifespan.
Understanding the clinical applications of the rigid bronchoscope—such as foreign body extraction, tumor debulking, stent placement, and control of massive hemoptysis—highlights the exposure to potentially infectious materials and the necessity for rigorous disinfection. Each use contaminates the instrument with microorganisms, organic matter, and biofilms, which, if not removed, can lead to device-related infections. Therefore, a structured cleaning regimen is not just a recommendation but a standard of care in respiratory medicine. In the following sections, we will outline the detailed cleaning流程 for rigid bronchoscopes, emphasizing the importance of each step in ensuring patient safety and device performance.
The cleaning process for a rigid bronchoscope begins immediately after its removal from the patient's airway. This initial step, known as pre-cleaning or point-of-use processing, is crucial for preventing the drying of organic material, which can make subsequent cleaning more difficult and less effective. According to standard protocols, the rigid bronchoscope should be wiped with wet gauze or a soft cloth to remove gross soil from its external surfaces . Additionally, any visible debris within the lumen should be flushed out using water or a enzymatic solution. This immediate action reduces the bioburden and minimizes the risk of biofilm formation, which can compromise disinfection later on.
During pre-cleaning, it is essential to handle the rigid bronchoscope with care to avoid damage to its optical and structural components. For instance, the distal tip and light post are delicate areas that require gentle wiping. If the rigid bronchoscope has detachable components, such as specific brands of video laryngoscopes , these should be disconnected according to the manufacturer's instructions to ensure all surfaces are accessible for cleaning. However, for most rigid bronchoscopes, the design is integral, so emphasis is placed on flushing and brushing the internal channels. After wiping and flushing, the instrument should be transported to the dedicated cleaning area in a closed container to prevent environmental contamination and protect it from physical damage .
Pre-cleaning also involves documenting the use of the rigid bronchoscope and any specific contamination concerns, such as exposure to tuberculosis or other highly resistant pathogens. This information will guide the subsequent disinfection steps. It is worth noting that pre-cleaning does not replace thorough cleaning and disinfection; rather, it is a preparatory step that enhances the efficacy of the overall process. In our OEM collaborations, we advise clients on designing rigid bronchoscopes with smooth, seamless surfaces and minimal joints to simplify pre-cleaning and reduce the risk of debris accumulation. This design philosophy aligns with the broader goal of infection prevention and device durability.
Manual cleaning is a foundational step in processing a rigid bronchoscope, as it physically removes contaminants that chemical disinfection alone cannot eliminate. This process should be conducted in a designated cleaning room equipped with a sink, brushes, enzymatic detergents, and personal protective equipment (PPE) for staff. The following steps outline a comprehensive manual cleaning approach for a rigid bronchoscope:
1. Leak Testing: Before immersion, perform a leak test to check for any damage to the rigid bronchoscope . This involves pressurizing the instrument and submerging it in water to detect air bubbles, which indicate leaks. If a leak is found, the device should be set aside for repair, as immersion could further damage internal components.
2. Disassembly: If the rigid bronchoscope has removable parts, such as valves or connectors, detach them according to the manufacturer's guidelines. This allows for thorough cleaning of each component. For example, some visual flexible laryngoscopes have detachable probes for easier cleaning .
3. Brushing and Flushing: Using a soft-bristled brush, scrub all accessible channels and the external surface of the rigid bronchoscope . Pay special attention to crevices and the distal tip. Then, flush the internal channels with water to remove loosened debris. Brushing should be done with compatible brushes that match the diameter of the channels to avoid damage.
4. Enzymatic Cleaning: Prepare an enzymatic detergent solution as per the product instructions. Immerse the rigid bronchoscope and all detached parts in this solution, and use a syringe or channel flusher to irrigate all lumens with the detergent . Enzymes break down organic materials like blood and mucus, enhancing contaminant removal. Gently agitate the solution to ensure full contact with all surfaces, and let it sit for the recommended time (usually 5-10 minutes).
5. Rinsing: After enzymatic cleaning, rinse the rigid bronchoscope thoroughly with clean water to remove any residual detergent . This step is critical, as leftover enzymatic agents can interfere with subsequent disinfection. Use forced air or water to flush out all channels, and wipe the exterior with a clean, lint-free cloth.
6. Inspection: Visually inspect the rigid bronchoscope under bright light for any remaining debris or damage. Use a magnifying glass if necessary. If contaminants are visible, repeat the brushing and rinsing steps. After inspection, dry the instrument with a soft cloth and proceed to disinfection.
Manual cleaning requires trained staff who understand the intricacies of the rigid bronchoscope and the importance of each step. In our OEM services, we provide detailed cleaning guidelines tailored to each device, ensuring compatibility with common enzymatic detergents and brushes. This support helps maintain the rigid bronchoscope's performance and safety throughout its lifecycle.
After manual cleaning, the rigid bronchoscope must undergo disinfection or sterilization to eliminate microorganisms. The choice between high-level disinfection (HLD) and sterilization depends on the intended use of the instrument. For example, rigid bronchoscopes that contact mucous membranes typically require HLD, while those used in sterile body sites may need sterilization. Common methods include chemical immersion and automated reprocessing, each with specific protocols.
Chemical disinfection involves immersing the rigid bronchoscope in an approved disinfectant solution. Commonly used agents include:
- Glutaraldehyde (e.g., 2% alkaline glutaraldehyde): This is effective for HLD and requires immersion for 20-45 minutes, depending on the pathogen load . For example, exposure to tuberculosis necessitates longer immersion times (at least 45 minutes). However, glutaraldehyde can irritate the respiratory system, so it should be used in well-ventilated areas with appropriate PPE.
- Ortho-phthalaldehyde (OPA): OPA is a faster-acting alternative that provides HLD in about 5 minutes . It has a low odor and does not require activation, but it may stain surfaces and cause allergic reactions in some individuals.
- Peracetic Acid and Hydrogen Peroxide-Based Solutions: These are used in automated endoscope reprocessors (AERs) and offer rapid, effective disinfection with minimal residue. They are suitable for rigid bronchoscopes that are compatible with AERs.
During chemical disinfection, ensure that the rigid bronchoscope is fully immersed, and all channels are filled with the disinfectant. After the contact time, remove the instrument and rinse it thoroughly with sterile water to eliminate chemical residues . This step is vital to prevent tissue irritation in patients. Finally, dry the rigid bronchoscope internally and externally before storage.
For rigid bronchoscopes used in sterile procedures, sterilization methods such as ethylene oxide (EO) gas or hydrogen peroxide plasma may be employed. EO sterilization is effective but requires a long cycle time and aeration to remove residual gas. Alternatively, low-temperature steam sterilization can be used for heat-sensitive components. Sterilization ensures the complete elimination of all microorganisms, including spores, but may not be necessary for all bronchoscopy procedures.
Automated endoscope reprocessors (AERs) are increasingly used for cleaning and disinfecting rigid bronchoscopes . These machines standardize the process by automating the flushing, disinfection, and rinsing steps, reducing the risk of human error. When using an AER, follow the manufacturer's instructions for connecting the rigid bronchoscope and selecting the appropriate cycle. AERs often integrate with bronchoscopy workstations and medical image processors to track cleaning cycles and maintain records for compliance.
In our OEM role, we design rigid bronchoscopes to be compatible with common disinfection methods and AERs. This includes using materials that withstand repeated exposure to chemicals and ensuring that all channels can be effectively flushed. By prioritizing cleanability, we help our clients deliver devices that meet regulatory standards and enhance patient safety.

Proper drying and storage are critical final steps in the cleaning process of a rigid bronchoscope. Inadequate drying can lead to microbial growth and biofilm formation in moist channels, compromising the device's safety for the next use. After disinfection and rinsing, the rigid bronchoscope should be thoroughly dried using forced air and clean, lint-free cloths.
- Drying Techniques: Use compressed air or automated drying systems to remove moisture from all internal channels of the rigid bronchoscope . Ensure that the air source is filtered and free of oil or contaminants. For external surfaces, wipe the instrument with a dry, soft cloth. Pay special attention to the distal tip and any crevices where water might accumulate. Some protocols recommend using 70-90% ethanol to flush the channels after drying, as it promotes evaporation and reduces the risk of waterborne pathogens . However, always check the manufacturer's guidelines to avoid material incompatibility.
- Storage Conditions: Once completely dry, the rigid bronchoscope should be stored in a dedicated, well-ventilated cabinet or room that protects it from dust, moisture, and physical damage . The storage area should be designed to prevent bending or stress on the instrument—for example, by hanging it vertically or placing it in a padded case. It is essential to ensure that the storage environment is clean and regularly disinfected to avoid recontamination. Additionally, label the rigid bronchoscope with the date of last cleaning and the next due date for maintenance, if applicable.
Proper storage not only preserves the rigid bronchoscope's condition but also extends its usable life. In our OEM services, we often advise on storage solutions that integrate with bronchoscopy workstations, such as customized racks or UV-disinfection cabinets. These innovations help maintain the readiness of the rigid bronchoscope for emergency procedures while upholding the highest standards of infection control.
Maintaining a rigid bronchoscope goes beyond cleaning; it involves regular quality control checks and preventive maintenance to ensure optimal performance and safety. Implement a tracking system for each rigid bronchoscope to log usage, cleaning cycles, and any issues encountered. This documentation aids in compliance with regulatory standards and facilitates timely repairs.
- Routine Inspections: Schedule periodic inspections for the rigid bronchoscope, focusing on the optical clarity, light transmission, and mechanical integrity. Check for scratches on the lens, dimming of the light source, or stiffness in the moving parts. Any abnormalities should be addressed immediately to avoid procedural delays or risks.
- Biological Monitoring: Conduct regular microbiological testing of the rigid bronchoscope and its channels to verify the effectiveness of the cleaning and disinfection processes . This involves swabbing internal channels and culturing the samples to detect any residual pathogens. If tests indicate contamination, review and reinforce cleaning protocols.
- Preventive Maintenance: Follow the manufacturer's recommended maintenance schedule for the rigid bronchoscope, which may include professional servicing, part replacements, or calibration. For instance, the light cables and seals may need periodic replacement to maintain performance. As an OEM provider, we offer maintenance support and accessories that ensure the longevity of our devices.
Incorporating these quality control measures into the workflow minimizes the risk of device-related infections and enhances the reliability of the rigid bronchoscope. Additionally, training staff on the latest cleaning protocols and technological updates is essential for consistent outcomes. Our company supports clients with comprehensive resources, including training modules and technical documentation, to uphold the highest standards in bronchoscope care.
Cleaning a rigid bronchoscope is a multifaceted process that demands attention to detail, adherence to protocols, and an understanding of the instrument's design. From immediate pre-cleaning to proper storage, each step plays a vital role in preventing infections and maintaining the device's functionality. As a leader in medical visualization technology, we are committed to advancing bronchoscope designs that simplify cleaning while delivering superior performance. Our OEM services empower brands and manufacturers to offer products that meet global hygiene standards, ultimately contributing to better patient outcomes. By following the guidelines outlined in this article, healthcare facilities can ensure the safe and effective use of rigid bronchoscopes in various clinical settings.
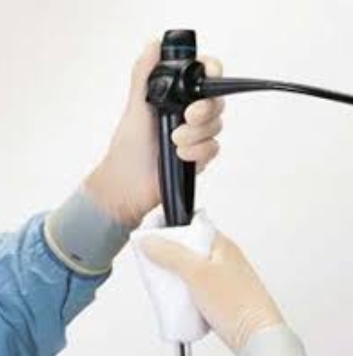
Manual cleaning is essential because it physically removes organic debris and biofilms that automated reprocessors might not fully eliminate . Automated systems are effective for disinfection but rely on prior manual cleaning to reduce bioburden. Skipping manual cleaning can lead to residual contamination and compromise patient safety.
Leak testing should be performed after each use of the rigid bronchoscope . This routine check helps identify early signs of damage, such as cracks or leaks, which could allow fluid infiltration and cause extensive internal damage. Regular leak testing extends the instrument's lifespan and ensures safety during procedures.
No, the choice of disinfectant depends on the bronchoscope's material compatibility and the manufacturer's instructions. For example, rigid bronchoscopes with optical components may require milder disinfectants to avoid damage, while others might tolerate stronger chemicals . Always refer to the device-specific guidelines to select an appropriate disinfectant.
A rigid bronchoscope should be stored in a dry, well-ventilated cabinet, preferably hung vertically or placed in a padded container to avoid physical stress . The storage area must be clean, disinfected regularly, and designed to prevent moisture accumulation, which can promote microbial growth.
The design significantly influences cleanability; for instance, rigid bronchoscopes with smooth surfaces, minimal joints, and accessible channels are easier to clean and disinfect . As an OEM provider, we focus on designing devices that simplify cleaning processes, thereby reducing infection risks and maintenance costs.
[1] https://v8.91huayi.com/cme/201212181515/02/jiangyi/content.html
[2] https://wap.cnki.net/touch/web/Journal/Article/SXLC200208047.html
[3] http://www.jmmedical.cn/news/i804953.html